Why Umbilical Wharton’s Jelly Mesenchymal Stem Cells?
Stem cell therapy is a broad and evolving field, with different types of stem cells sourced from various tissues. When exploring stem cell therapy services in Johor Bahru, patients often discover that there is no single stem cell source used across all clinics. Each source differs in terms of origin, preparation process, and clinical considerations.
Because of these differences, clinics must carefully evaluate which stem cell source best aligns with safety standards, patient comfort, and consistency in processing. Among the many available options, umbilical Wharton’s jelly mesenchymal stem cells are chosen based on these factors. In addition, a recent search on ClinicalTrials.gov shows more than 60 registered clinical trials that reference Wharton’s jelly, reflecting ongoing research interest in this stem cell source across various medical fields.
Clinical trials listed on ClinicalTrials.gov represent ongoing or completed research studies and do not imply regulatory approval or guaranteed outcomes.
Understanding Umbilical Wharton’s Jelly
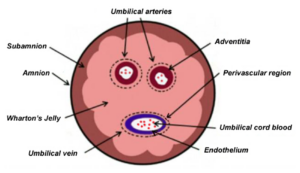
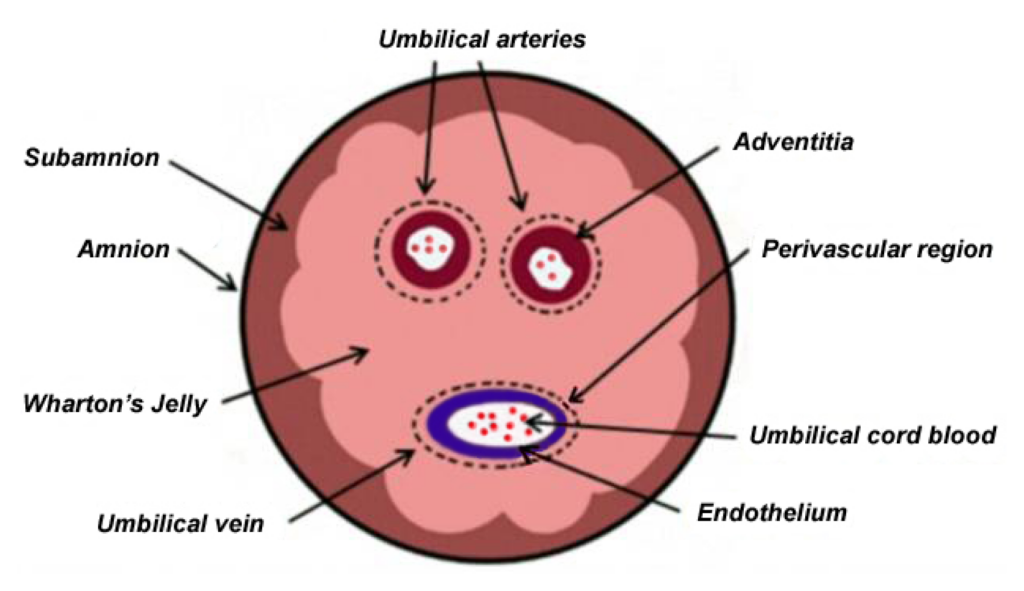
Wharton’s jelly is a soft, protective tissue found within the umbilical cord. Its natural role is to cushion and protect the blood vessels during pregnancy. This tissue is rich in mesenchymal stem cells, which are widely studied for their ability to support the body’s natural repair and regulatory processes.
The umbilical cord is collected after childbirth, making the sourcing process non-invasive and ethically acceptable. No procedure is performed on the patient or donor for collection.
Five Key Reasons Umbilical Wharton’s Jelly MSCs Are Used
1. Multipotent Properties
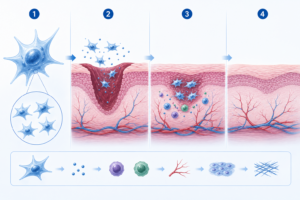
Umbilical Wharton’s jelly mesenchymal stem cells are considered multipotent, meaning they are studied for their ability to develop into different types of specialised cells. This characteristic is one of the reasons they are widely researched and discussed in regenerative medicine, particularly in relation to tissue support and repair processes.
2. Immune Response Considerations
These cells are often described as having immune-modulating characteristics, meaning they are less likely to trigger strong immune reactions when compared with some other stem cell sources. This makes them suitable for broader clinical consideration, subject to proper medical assessment.
3. Observed Wellness Support
In clinical settings, patients receiving stem cell-related services using umbilical cord MSCs have reported improvements in overall wellbeing. Individual responses vary, and outcomes depend on many factors, including health condition and medical history.
4. Patient Comfort
Unlike stem cells collected from bone marrow or fat tissue, umbilical Wharton’s jelly MSCs do not require invasive harvesting procedures from the patient. This helps reduce discomfort, procedural risks, and recovery time associated with cell collection.
5. Ethical Sourcing and Practical Availability
Umbilical cords are collected after childbirth through an ethical and non-invasive process. With proper screening and controlled laboratory processing, this source offers consistent availability while supporting sustainability and safety standards.
Important Note
Stem cell-related services are not a cure and are not suitable for everyone. The use of umbilical Wharton’s jelly mesenchymal stem cells is based on clinical considerations, safety protocols, and regulatory guidance. A medical consultation is essential to determine suitability on an individual basis.
Patients are encouraged to consult their medical professional or book an appointment with our clinic to discuss whether this approach may be appropriate for their individual needs.